Cartridge Information
Our cartridges are developed for research-use environments and engineered to support consistent, repeatable delivery with low residual volume. The design combines precision-formed internal geometry with a long-stopper system that travels further into the neck of the cartridge, helping to evacuate fluid efficiently.
They are designed as a low dead space, high-recovery system, supporting efficient fluid delivery and minimal retained volume.
Typical residual volume is approximately 0.03–0.05 mL in a 3 mL cartridge, supporting consistent delivery across batches. As with all cartridge and syringe systems, a small amount of residual volume is expected. Our cartridges are designed to operate within accepted performance ranges, delivering consistent results in real-world use recognized injection system standards, including ISO 7886 and ISO 11608, which focus on dose accuracy and repeatable delivery.
How are the 3 mL cartridges sterilized?
For both of our 3 mL cartridge models, individual components are steam sterilized prior to assembly. Components are then mechanically assembled in a controlled sterile environment, sealed in Tyvek steam sterilization pouches and then a final steam sterilization. Each pouch includes a visual steam indicator confirming proper exposure to sterilization conditions.
Are the cartridges sterilized?
Cartridges are assembled and undergo a final steam sterilization cycle after packaging. Pen-Depot.com often sells multi-cartridge pouched items with 4-10 cartridges in the same steam sterilized pouch as well as individually wrapped cartridges,
What type of packaging is used?
The cartridges are sealed in a Tyvek steam sterilization pouch. Tyvek is widely used for steam sterilization due to its durability, breathability, and ability to maintain package integrity following autoclave processing.
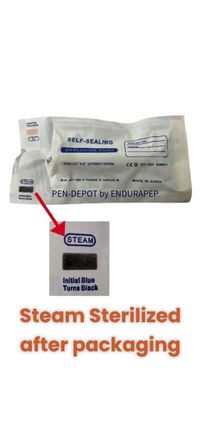
How can I tell the product has been steam sterilized?
Each sealed pouch includes a steam-activated visual indicator. This indicator changes color from Blue to shades of black or green depending on the pouch used when appropriate temperature and steam exposure conditions have been met during the autoclave cycle, This provides clear visual confirmation of processing.
Do you provide sterilization certificates, validation logs, or testing reports?
No. We do not provide individual sterilization certificates, batch records, biological indicator results, or testing reports due to the sheer volume of items sold.
Verification is provided solely by the visual steam indicator on the sealed pouch.
Our products are not marketed or classified as regulated medical devices. The sterilization process described reflects quality-controlled handling appropriate for the product’s intended classification and use.
Why don’t you offer additional sterilization documentation?
Individual certification and validation reporting is typically reserved for regulated medical or surgical devices. For our research product category, visual indicator verification on sealed sterilization pouches is the accepted standard.
How do I know if this product is right for my needs?
Customers are encouraged to assess suitability based on their own research requirements and intended use prior to purchase.
If additional documentation or regulatory validation is required for a specific research use case, this product may not be the appropriate fit.
